Learn the tests doctors use—symptoms, TVUS, biopsy, MRI—and how fast diagnosis works in Spain. Book a same-week consultation with Dr. Lucas Minig.
Introduction
Endometrial cancer — cancer of the uterine lining — most commonly presents with abnormal uterine bleeding and is usually diagnosed after a short diagnostic pathway that combines clinical assessment, imaging and, crucially, tissue sampling. This article walks through each step of that process in practical, patient-facing language, explains what tests show and why they matter, and highlights how care is organized in Spain and in surgeon-led, minimally invasive units such as the one led by Dr. Lucas Minig.
Why prompt diagnosis matters
Early detection of endometrial cancer is important because most tumors confined to the uterine lining can be treated curatively with surgery. Delays in diagnosis increase the chance the tumor will invade the muscle of the uterus or spread beyond it, which can require more extensive treatment. Acting quickly after first warning signs improves outcomes and often simplifies the treatment plan.
Outcomes improve with early detection
When endometrial cancer is found while still limited to the uterus, five-year survival rates are high compared with later stages. That’s why clinicians prioritize rapid evaluation of abnormal bleeding: small delays can mean a shift in stage and therefore a different treatment intensity.
Common warning sign: abnormal bleeding
The hallmark symptom that triggers investigation is abnormal vaginal bleeding — especially bleeding after menopause, spotting between periods, or heavier menstrual bleeding than usual. If you’re postmenopausal and notice any bleeding, clinical assessment should be prompt.
How Is Endometrial Cancer Diagnosed?
Diagnosis is a stepwise process:
- Clinical assessment (history + pelvic exam)
- First-line imaging (usually transvaginal ultrasound)
- Tissue diagnosis (endometrial sampling via Pipelle, hysteroscopy or D&C)
- Detailed pathology (histology, grade and molecular testing)
- Staging scans (MRI + selected CT/chest imaging)
This layered approach ensures both accurate diagnosis and the information needed to plan the right treatment.
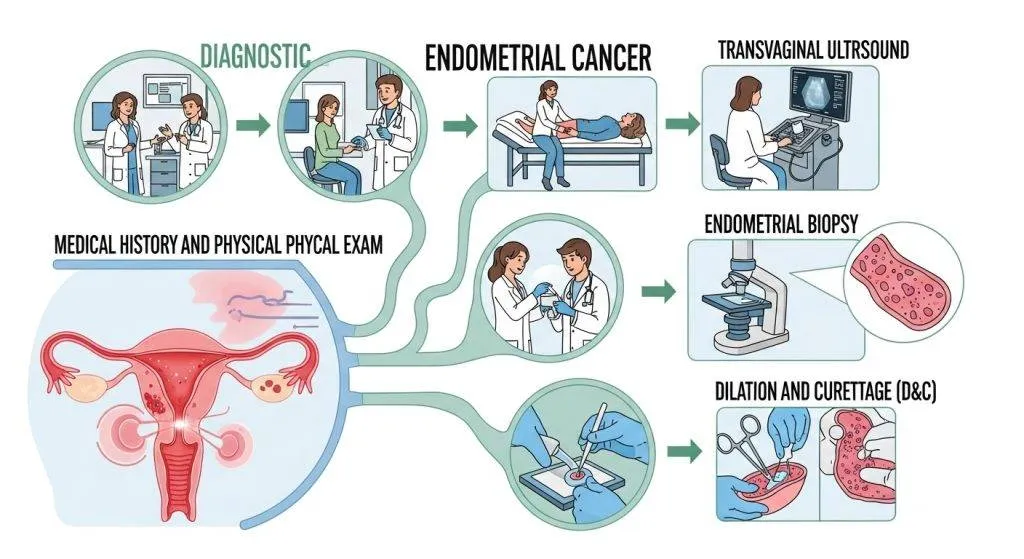
Step 1: Clinical assessment (history & pelvic exam)
A careful history focuses on the bleeding pattern, menopausal status, use of hormonal therapy, medications, and systemic risk factors (obesity, diabetes, unopposed estrogen exposure, family cancer syndromes). The pelvic exam can detect obvious pelvic masses or lesions but cannot confirm or rule out endometrial cancer by itself. The history and exam set the urgency and the choice of the next test.
What doctors ask and why it matters
Typical questions: When did bleeding start? How heavy is it? Any pain? Prior gynecologic history? Family history of colorectal or endometrial cancer (which may raise concern for Lynch syndrome). These details influence whether an immediate biopsy or an ultrasound-first approach is best.
Risk factors that raise suspicion
Risk factors that increase suspicion include age (postmenopausal), obesity, polycystic ovary syndrome, long-term tamoxifen use, and a personal or family history suggesting hereditary cancer syndromes such as Lynch syndrome. Patients with these features often move more quickly to tissue sampling.
Step 2: Transvaginal ultrasound (TVUS) — first imaging test
Transvaginal ultrasound (TVUS) is the usual first-line imaging study because it is noninvasive, quick and widely available. The sonographer evaluates endometrial thickness, the presence of focal lesions (polyps or masses) and intrauterine fluid, all of which guide further testing.
What the ultrasound shows (endometrial thickness, polyps, fluid)
- Endometrial thickness: a thin lining in a postmenopausal woman is usually reassuring; a thicker-than-expected lining prompts biopsy.
- Polyps or focal masses: may be visible and can be targeted later with hysteroscopy.
- Fluid in the uterine cavity: can be associated with pathology when seen in a postmenopausal patient and typically warrants sampling.
Endometrial thickness thresholds: quick rules
Clinical practice often uses thresholds to decide next steps: for example, an endometrial thickness >4–5 mm in a postmenopausal woman with bleeding commonly prompts tissue sampling. Thresholds vary slightly by guideline and clinical context, so clinicians combine ultrasound results with symptoms and risk factors to decide.
Step 3: Tissue diagnosis — biopsy methods compared
A tissue sample is required to confirm endometrial cancer. There are three main approaches: office (Pipelle) biopsy, hysteroscopy with directed biopsy, and dilation & curettage (D&C). Each has strengths and trade-offs.
Office endometrial biopsy (Pipelle)
The Pipelle is a narrow suction catheter used in the outpatient clinic to take a small sample of the uterine lining. It’s quick, generally well tolerated, and is the first-line sampling method in many settings because it often provides enough tissue to diagnose cancer without anesthesia. Its diagnostic yield is high, especially in postmenopausal women with bleeding.
Hysteroscopy with directed biopsy
Hysteroscopy allows direct visualization of the uterine cavity with targeted biopsies of suspicious areas (for example, a polyp or focal lesion). This increases diagnostic accuracy when the ultrasound shows a focal lesion or when prior blind sampling is inconclusive. Newer practice favors office hysteroscopy when possible because it combines diagnosis and treatment (polyp removal) in the same procedure.
D&C (dilation & curettage): when it’s used
D&C is performed under anesthesia and involves scraping the uterine lining; it’s reserved when office sampling and hysteroscopic sampling are not feasible or if a larger tissue sample is required. Although less commonly the first choice, D&C remains a valid diagnostic tool in specific situations.
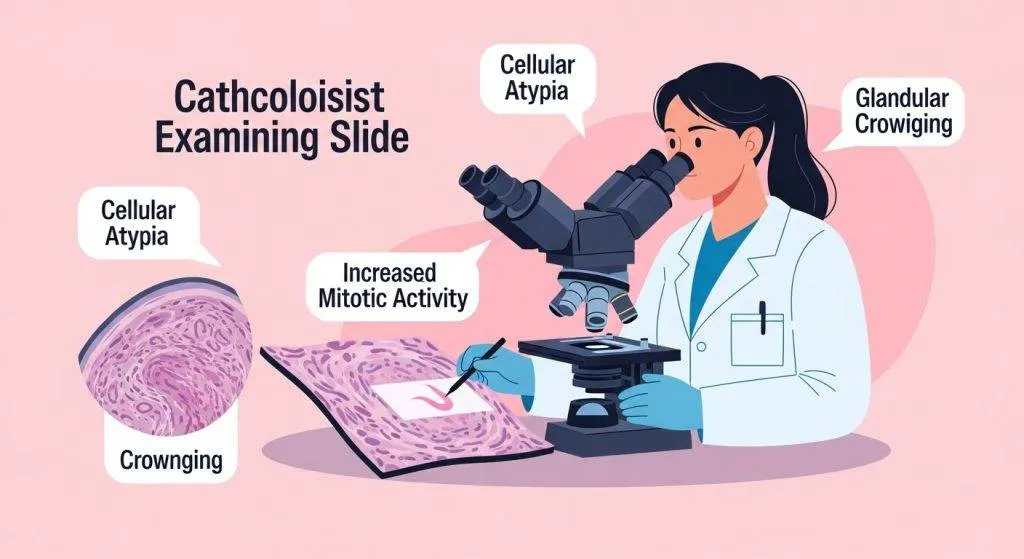
Step 4: Pathology report — beyond “cancer”
A pathology report goes beyond simply reporting cancer; it specifies the histologic subtype (endometrioid, serous, clear cell, etc.), the tumor grade (how abnormal the cells look), and increasingly, molecular characteristics that influence prognosis and treatment selection.
Histologic type and grade explained simply
- Histologic type: tells which kind of endometrial cancer the tumor is (endometrioid is the most common).
- Grade: ranges from low (well-differentiated) to high (poorly differentiated) and predicts how aggressive the tumor behavior may be. These features factor into surgical planning.
Molecular testing: MMR, Lynch, TCGA groups
Modern pathology increasingly includes molecular tests: mismatch-repair (MMR) protein immunohistochemistry (to screen for Lynch syndrome), and molecular classification (TCGA-informed groups) that refine prognosis and help personalize adjuvant therapy. Many specialist centers in Spain already include these tests as part of the diagnostic report.
Step 5: Staging scans — MRI, CT, chest imaging
Once cancer is confirmed, imaging maps local extent and potential spread.
Why MRI is preferred for local staging
MRI is the gold standard for assessing how deeply the tumor invades the myometrium and whether the cervix is involved — information that determines surgical approach and the need for lymph node assessment. MRI protocols tailored to endometrial cancer (including diffusion sequences) improve accuracy.
When CT or chest imaging is indicated
CT scans (abdomen/pelvis) or chest imaging are used to evaluate lymph nodes or distant spread when clinically indicated. PET/CT is reserved for selected high-risk cases. Choice of scan is individualized based on pathology, symptoms and local protocols.
Common diagnostic pathways and timelines
In specialist clinics, a “fast-track” pathway can move a patient from first visit to biopsy and pathology result in days to a couple of weeks. Public systems vary by region; private specialist units often offer rapid coordinated care combining ultrasound, office biopsy or hysteroscopy and early multidisciplinary review. Rapid pathways reduce anxiety and allow timely treatment planning.
Typical fast-track timeline in a specialist clinic
Example timeline: Day 0 — initial consult and TVUS; Day 1–7 — office Pipelle or hysteroscopy; Day 7–14 — pathology report and MRI staging if needed; Days 14–28 — multidisciplinary review and surgical planning. Timelines differ by center and region.
How care looks different in specialist hands
Specialist gynecologic oncologists and multidisciplinary teams bring the advantage of experience in minimally invasive surgery, modern staging protocols and integrated molecular reporting. Surgeon-led units that prioritize minimally invasive and fertility-sparing options (when appropriate) can often offer tailored, less morbid treatment pathways. Units led by experienced surgeons such as Dr. Lucas Minig emphasize rapid coordinated diagnostics and minimally invasive surgical techniques.
Diagnosis and decisions: what the results mean for treatment
Diagnostic results determine the next steps: a low-grade, early tumor may be treated with hysterectomy alone (often minimally invasive), while high-grade or deeply invasive tumors may require lymph node assessment and adjuvant therapies (radiation or chemotherapy). Molecular results are increasingly used to refine adjuvant therapy decisions.
Diagnosis pathway in Spain
Spain combines national society guidance with regional health system pathways. Public hospitals follow oncoguías and regional tumor boards, while private specialist centers offer rapid, coordinated services including office biopsy, hysteroscopy and prompt molecular testing. Patients should confirm with their clinic whether molecular profiling and prompt MRI staging are included, and discuss referral to a gynecologic oncologist for confirmed cancers.
Meet the doctor: surgeon-led, minimally invasive care — Dr. Lucas Minig
If you prefer a surgeon-led, minimally invasive approach with coordinated diagnostics, units like Dr. Lucas Minig’s emphasize same-week evaluation, high rates of laparoscopic/robotic surgery and multidisciplinary decision-making. These features can shorten the pathway from symptom to definitive treatment and often reduce recovery time after surgery. Ask your doctor about experience, yearly case volume and whether the unit performs molecular reporting as part of the routine pathology.
Conclusion
Diagnosing endometrial cancer is a structured process: clinical assessment prompts ultrasound, which guides tissue sampling (Pipelle, hysteroscopy or D&C). Pathology — including molecular testing — combined with MRI staging provides the roadmap for treatment. Prompt evaluation of abnormal bleeding, especially after menopause, remains the most important patient action. In Spain, coordinated specialist teams and private surgeon-led units can offer rapid, tailored diagnostic and treatment pathways.
FAQs
Can a Pap smear detect endometrial cancer?
No — Pap tests screen the cervix and are not reliable for endometrial disease; any abnormal bleeding warrants specific evaluation for the endometrium.
Is the office biopsy painful?
Most patients report brief cramping; it’s short and typically well tolerated without general anesthesia.
How long until I get results?
Routine pathology usually returns in 5–10 working days; molecular panels may add several days to a couple of weeks.
What if the Pipelle is negative but I still have bleeding?
Persistent bleeding after a negative office biopsy usually leads to hysteroscopy with directed biopsy or D&C to rule out focal lesions.
If I’m in Spain, how quickly can I be evaluated?
Many Spanish specialist clinics offer teleconsults and fast-track imaging/biopsy; surgeon-led practices can often provide a complete diagnostic pathway within 2–4 weeks, faster in urgent cases.