Learn whether endometrial cancer spreads in weeks, months, or years — clear timelines, aggressive vs slow types, red flags, and what to do next. Fast specialist help available in Spain.
Introduction
If you’re asking “how fast does endometrial cancer spread?”, you’re asking the right question — and the answer isn’t a single number. Speed depends on the tumor’s type, grade, and depth of invasion, as well as individual health factors. This short guide cuts through the medical jargon to explain which cancers tend to progress slowly, which behave aggressively, what warning symptoms to watch for, and how specialists (including teams in Spain and experienced clinicians like Dr. Lucas Minig) work quickly to diagnose and treat patients so spread is caught early and outcomes are optimized.
How Fast Does Endometrial Cancer Spread?
There is no single clock for every patient — speed depends on tumour biology, grade, and depth of invasion. Read on to understand the main patterns and what they mean for timing of diagnosis and treatment.
Slow-growing vs aggressive types
- Slow-growing (typical “type I” endometrioid): These tumours often develop in association with estrogen exposure and are commonly low-grade. They frequently cause abnormal bleeding early, which prompts testing. Because they’re discovered earlier and their cells divide more slowly, many remain localized for months or even years.
- Aggressive types (typical “type II” — serous or clear-cell, and high-grade tumours): These are biologically different — they may appear with fewer warning symptoms and have a higher tendency for deep invasion and early spread to lymph nodes or other organs. In such cases, spread can occur within weeks to months if left untreated.
Practical takeaway: the phrase “how fast” is meaningful only when tied to the tumour’s type and grade.
Why there is no universal timeline
Tumour growth and spread is influenced by microscopic features (grade), molecular drivers, host factors (immune response, comorbidities), and timing of intervention. Two patients with the same size lesion on ultrasound can have very different outcomes because their cancers may behave differently at the cellular level. That variability makes precise timelines impossible — clinicians therefore describe risk categories (low, intermediate, high) and act accordingly.
What “speed of spread” really means (biology, grade, and stage)
Broadly, “speed” refers to how quickly cancer moves from the uterine lining into muscle, nearby tissues, lymph nodes, and distant organs. Clinicians translate this into grade (cell appearance) and stage (how far the cancer has already traveled).
Tumor grade (1–3): how aggressive are the cells?
- Grade 1: Cells look similar to normal endometrium — usually slower growing.
- Grade 2: Moderately abnormal.
- Grade 3: Highly atypical cells that replicate quickly and are likelier to spread.
Grade matters because high-grade tumors are biologically primed to progress faster.
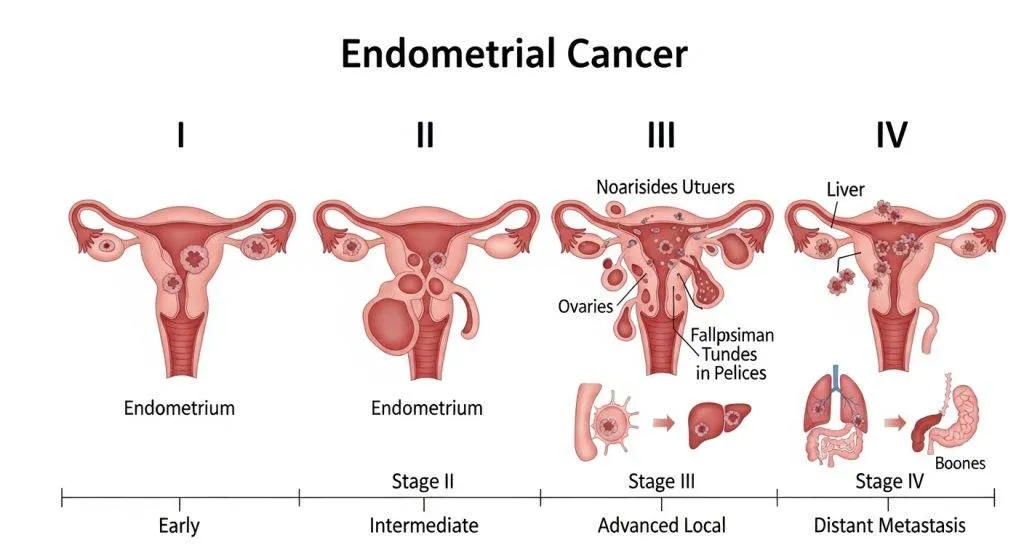
Tumor stage: how far has it already gone?
- Stage I: Confined to the uterus.
- Stage II: Reaches the cervix.
- Stage III: Extended to pelvic tissues or lymph nodes.
- Stage IV: Distant spread (lungs, liver, peritoneum).
Stage is the main prognostic marker: the higher the stage at diagnosis, the more aggressive the treatment needed.
Myometrial invasion and LVSI (why depth matters)
Two key pathological features predict spread risk:
- Depth of myometrial invasion: The deeper the tumor penetrates the uterine muscle, the higher the chance cells reach lymphatics.
- Lymphovascular space invasion (LVSI): Cancer cells seen inside blood or lymph vessels is a red flag for early dissemination.
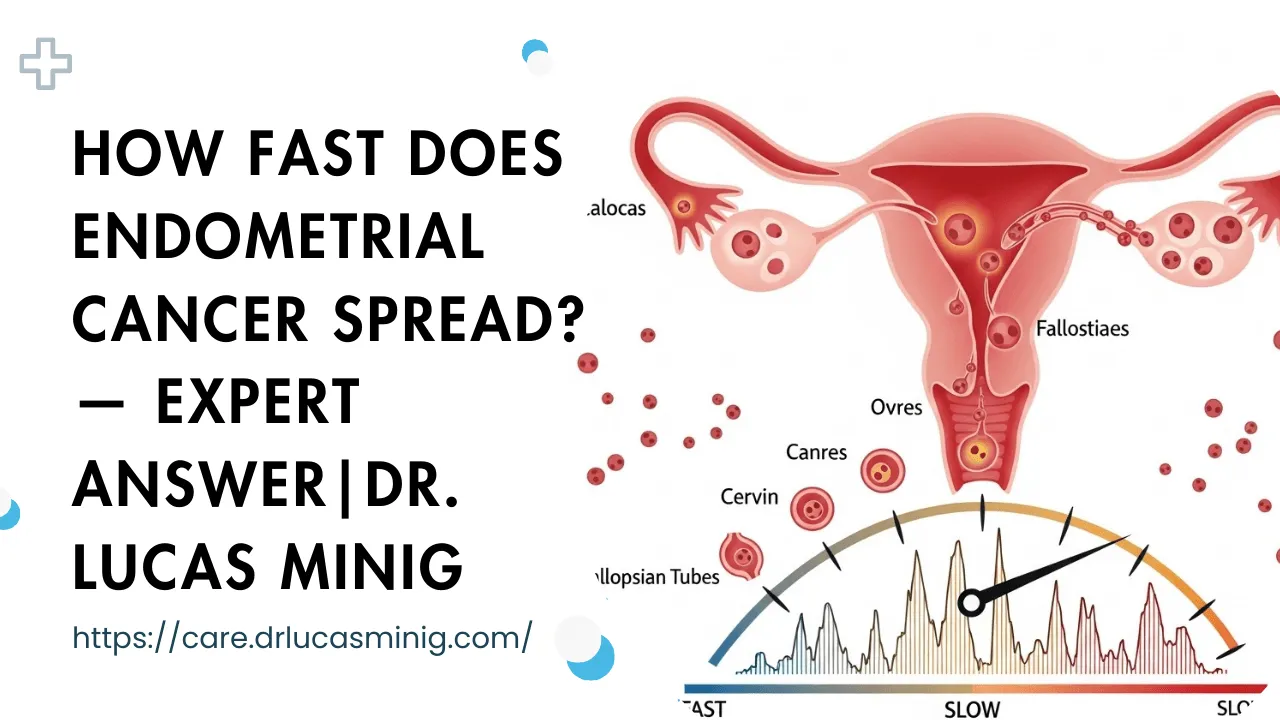
Common pathways of spread
Endometrial cancer uses predictable anatomic routes: local extension, lymphatic channels to nodes, peritoneal surfaces, and — less commonly — bloodborne routes to distant organs. Understanding these helps guide imaging and surgical planning.
Local extension (uterus → cervix → pelvis)
Initial growth often extends from the uterine lining into the myometrium and then overtly into the cervix or adjacent pelvic tissues. Local extension increases the complexity of surgery and raises the likelihood of needing additional therapies.
Lymphatic spread (pelvic and para-aortic nodes)
Tumour cells frequently travel through pelvic lymphatics to pelvic and para-aortic nodes. Node involvement may be microscopic (detected only with sentinel node biopsy or pathology) or macroscopic on imaging. Presence of nodal disease shifts staging and typically requires multimodal therapy.
Peritoneal seeding and distant metastases (lungs, liver)
In some tumours, cells shed into the uterine cavity or peritoneal fluid and implant on pelvic surfaces; in others, hematogenous spread seeds distant organs — most commonly lungs, then liver and bone. Distant metastases represent advanced disease and are typically associated with a more rapid clinical course.
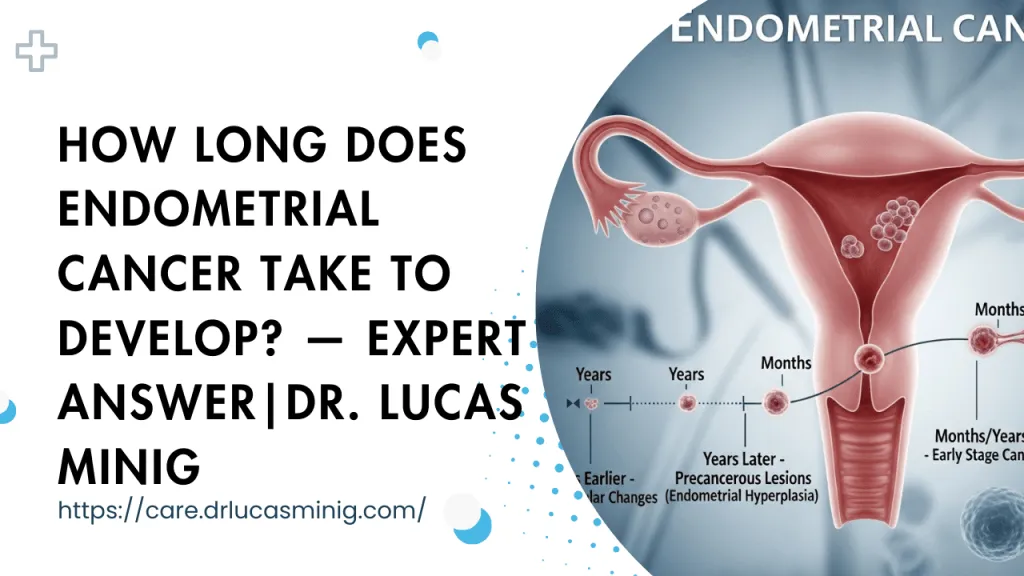
Typical timelines: weeks, months, or years?
While every case is unique, clinicians use illustrative timelines that reflect average patterns seen in clinical practice.
Typical low-grade (Type I) scenario
A low-grade endometrioid tumour often presents with abnormal bleeding, is biopsied and treated within weeks. If untreated (rare once symptomatic), progression from localized disease to regionally advanced disease may take many months or years in many cases — which is why timely diagnosis leads to excellent outcomes.
Aggressive (Type II / high-grade) scenario
High-grade serous or clear-cell cancers can progress rapidly. In a small but important subset, these tumours present already with regional or distant spread; without intervention they can show measurable progression over weeks to a few months.
Examples from clinical studies (recurrence / progression)
Clinical literature consistently shows that recurrence patterns and time to progression are strongly correlated with stage and grade: low-stage, low-grade cancers have low recurrence rates and long disease-free intervals; high-grade or advanced-stage cancers recur sooner and more often. (These study data inform risk-stratified follow-up schedules.)
Which factors make it spread faster?
Some features reliably increase the risk of faster spread — knowing them helps doctors choose more aggressive staging and therapy.
Histologic subtype (serous, clear cell)
These subtypes are inherently more aggressive and carry higher rates of extrauterine disease at diagnosis.
High grade, deep myometrial invasion, LVSI
The combination of high grade + deep invasion + LVSI is a red flag: it significantly raises the probability of nodal and distant involvement and shortens time to progression in untreated scenarios.
Molecular markers and patient factors (age, obesity, diabetes)
Molecular profiling (p53 abnormalities, mismatch repair deficiency, POLE mutations) increasingly refines risk — some mutations predict faster behaviour. Patient factors like obesity and diabetes are linked to incidence and may affect presentation and comorbidity but are less directly predictive of short-term spread than tumour biology.
Symptoms That May Signal Faster Spread
Certain new or worsening symptoms suggest the cancer may be more advanced or spreading — prompt assessment is essential.
Abnormal bleeding
Postmenopausal bleeding or any abnormal uterine bleeding is the most common early symptom. New or heavy bleeding can indicate tumor growth; persistent bleeding requires immediate evaluation.
Pelvic pain and pressure
Increasing pelvic pain, a palpable mass, or pressure symptoms (urinary frequency, bowel changes) may indicate local extension beyond the uterus.
Systemic symptoms
Unexplained weight loss, breathlessness, or persistent cough can flag distant spread (e.g., to the lungs) and warrant urgent imaging.
How doctors in Spain (and worldwide) assess spread
Assessment combines imaging, endometrial sampling, and surgical evaluation — the goal is accurate staging with minimal morbidity.
Imaging: ultrasound, MRI, CT and PET-CT
- Transvaginal ultrasound: first-line for thickness and structural assessment.
- Pelvic MRI: best for evaluating myometrial invasion and cervical involvement.
- CT chest/abdomen or PET-CT: used to detect nodal enlargement or distant disease when risk is higher or symptoms suggest spread.
Biopsy, hysteroscopy and pathology report
Endometrial biopsy (or hysteroscopy–directed sampling) provides diagnosis and grade. The pathology report will comment on grade, LVSI, and other features that guide staging and treatment.
Sentinel lymph node vs full lymphadenectomy
Modern practice increasingly uses sentinel lymph node mapping to detect nodal disease with less surgical morbidity; full lymphadenectomy is reserved for select high-risk cases. The choice depends on preoperative risk assessment and intraoperative findings.
(As Dr. Lucas Minig and many gynecologic oncology teams explain, timely, accurate staging shortens the pathway to definitive treatment.)
Treatment quickly changes the picture — why early action matters
Proper treatment can stop progression — surgery often removes the primary source and adjuvant therapies treat microscopic disease that could seed elsewhere.
Surgery is often first line (hysterectomy ± nodes)
Standard care for early disease is total hysterectomy with removal of fallopian tubes and often ovaries, plus assessment of lymph nodes as indicated. Minimally invasive approaches reduce recovery time without compromising oncologic outcomes in appropriate cases.
Adjuvant therapy for higher risk disease (radiation, chemo, immunotherapy)
For high-risk or node-positive disease, adjuvant radiation and/or chemotherapy reduces recurrence risk. Emerging treatments, including targeted therapies and immunotherapy guided by molecular markers, are changing outcomes for some patients.
What to do if you notice symptoms (in Spain and elsewhere)
Don’t delay — abnormal bleeding or new pelvic symptoms need prompt medical review and a clear referral to gynecology.
Abnormal bleeding — urgency and referral pathways
In many clinical systems, abnormal postmenopausal bleeding triggers fast-track referral to gynecology for ultrasound and biopsy. If you are in the country and experience such symptoms, ask your GP for a prioritized gynecology appointment or contact a specialist clinic directly.
What your initial consult will look like (questions doctors ask)
Expect questions about bleeding history, menopause status, medications, family cancer history, and general health. The clinician will usually arrange ultrasound and a biopsy if indicated and explain next steps for staging and management.
Prognosis: survival and outcome depend on stage at diagnosis
A concise lead: Early-stage, low-grade cancers have excellent prognosis; survival declines with nodal or distant spread. That’s why staging and timely treatment are so powerful predictors of outcome.
How Dr. Lucas and specialist team speed up diagnosis and care
Experienced teams — surgeons, radiologists, pathologists, and medical/radiation oncologists — streamline triage: rapid imaging, targeted biopsy, sentinel node mapping, and a coordinated surgical plan minimize delays and operative morbidity. In private and specialist centers across Spain, streamlined pathways (fast teleconsults, rapid slot imaging, and same-week surgical planning) shorten the window between symptoms and definitive treatment, reducing the chance of further spread.
Common myths and honest answers
A short lead: Misinformation creates fear. Here are direct corrections to common myths.
- Myth: “All uterine cancers spread rapidly.” — Truth: Many do not; behaviour depends on biology.
- Myth: “If I have bleeding I definitely have cancer.” — Truth: Bleeding has many causes; still, it always deserves assessment.
- Myth: “Surgery will always be delayed.” — Truth: Many centres prioritize suspected cancers; ask for urgent pathways.
Conclusion
Endometrial cancer’s speed of spread is not one-size-fits-all. It is governed by tumour type, grade, myometrial invasion, LVSI, and molecular features. Many cases are slow-growing and curable with prompt treatment; aggressive subtypes demand swift, coordinated care. If you notice abnormal bleeding or new pelvic symptoms, seek evaluation — early action makes a measurable difference in outcomes.
FAQs
How quickly should I see a doctor for abnormal uterine bleeding?
Seek assessment promptly — ideally within days to weeks, because ultrasound and biopsy can usually be arranged quickly and provide essential answers.
Can a low-grade endometrial cancer become aggressive if untreated?
While progression is possible, low-grade tumours generally advance slowly; nonetheless, timely removal is recommended because individual behaviour can vary.
Are there blood tests that tell how fast the cancer is spreading?
No single blood test measures spread speed. Tests (imaging and pathology) and markers may suggest extent and risk, but the definitive assessment is tissue diagnosis and staging.
What role does obesity play in endometrial cancer behaviour?
Obesity is a risk factor for developing many endometrioid endometrial cancers and can influence overall health and treatment options — but it’s not the sole determinant of how fast a tumour spreads.
Where can I get a specialist opinion quickly?
Ask your GP for expedited referral to a gynecologic oncology team or contact a specialized clinic directly; many centres offer rapid assessment and teleconsultations.