Discover what studies and Spanish gynecologists say about Mirena (LNG-IUS) and endometrial cancer risk. Clear, evidence-based guidance from Dr. Lucas Minig.
Introduction
Mirena—the levonorgestrel intrauterine system (LNG-IUS)—is a common question topic for women worried about uterine cancer. Patients ask whether this IUS actually prevents endometrial cancer or merely lowers the risk.
Below you’ll find a practical, clinic-minded explanation written for people in Spain and for doctors like Dr. Lucas who counsel patients about options. Read on for clear takeaways and realistic expectations.
What is Mirena?
Mirena is a small T-shaped intrauterine device that slowly releases levonorgestrel directly into the uterus. It’s used worldwide for contraception and to treat heavy menstrual bleeding, and it’s commonly prescribed in Spain.
This local delivery produces strong effects on the endometrium with relatively low systemic hormone levels, which makes Mirena different from many oral hormones.
How the Mirena IUS works
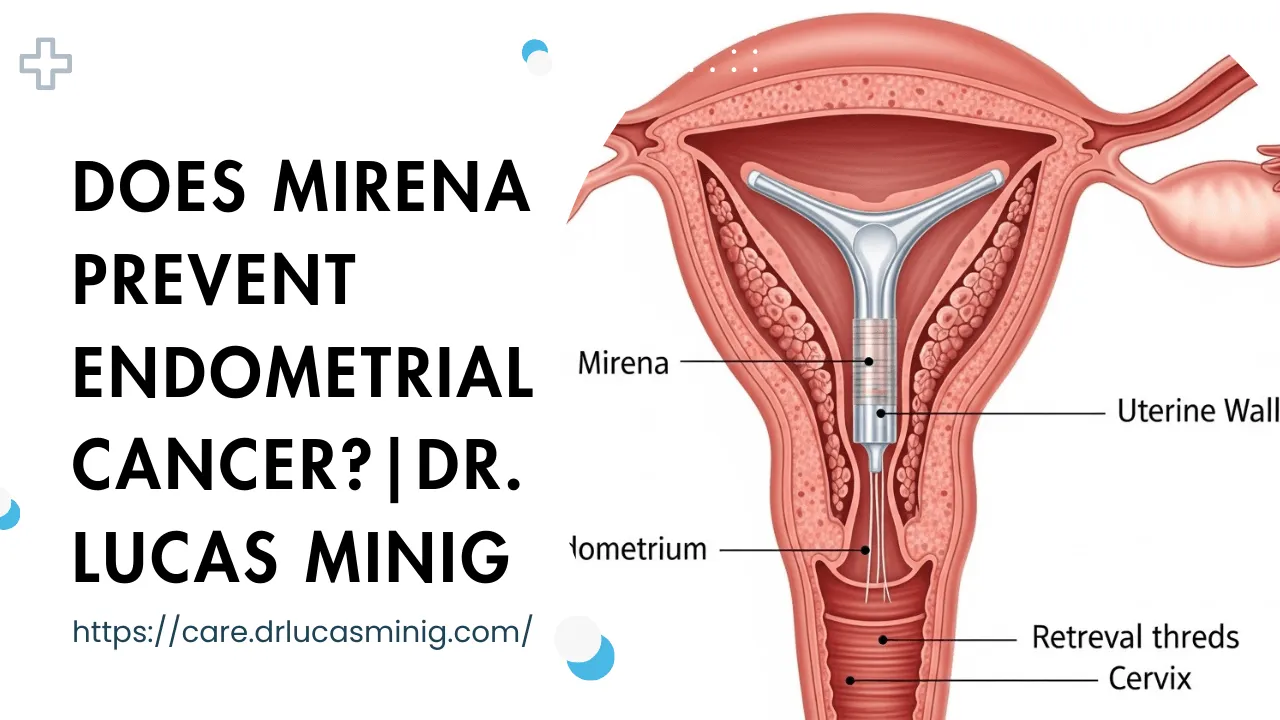
Mirena sits inside the uterine cavity and steadily releases levonorgestrel over several years. The hormone acts primarily on the endometrium (the uterine lining), suppressing its growth and changing the local environment to make implantation and heavy bleeding less likely.
Hormone released: levonorgestrel explained
Levonorgestrel is a progestin — a synthetic form of progesterone. It counteracts estrogen’s stimulating effect on the uterine lining, causing thinning (atrophy) and reduced bleeding. Because it’s delivered locally, the uterus sees higher concentrations than the rest of the body.
Licensed uses of Mirena in Spain (AEMPS/CIMA)
In Spain Mirena is approved for long-acting contraception and for treating heavy menstrual bleeding. Clinicians also use it within protocols for endometrial hyperplasia and, selectively, as part of fertility-sparing care — always following national guidance and specialist judgment.
Does Mirena prevent endometrial cancer?
Patients frequently ask: does Mirena prevent endometrial cancer? The short, honest answer is that Mirena is associated with a reduced risk of endometrial cancer, but it should not be called an absolute prevention tool.
Evidence comes largely from observational studies and meta-analyses showing lower cancer incidence among LNG-IUS users, yet randomized prevention trials specifically designed to prove “prevention” are lacking.
What research suggests (cohort studies, meta-analyses)
Large cohort studies and pooled analyses show a consistent association between LNG-IUS use and lower rates of endometrial hyperplasia and cancer. Some studies report substantial relative risk reductions, particularly in groups with higher baseline risk. These data support the idea that Mirena creates an unfavorable environment for precancerous changes.
Why doctors say “risk reduction” rather than “prevention”
There are three reasons clinicians prefer the phrase risk reduction:
- Most high-quality data are observational (real-world cohorts), which show association but cannot definitively prove causation.
- There are no large randomized clinical trials designed and powered specifically to test Mirena as a primary preventive intervention for endometrial cancer in the general population.
- Individual risk depends on many factors (genetics, obesity, exogenous hormones, age), so an IUS lowers one major mechanism (unopposed estrogen) but does not eliminate other risks.
So clinicians in Spain and elsewhere accurately describe Mirena as offering protection by reducing risk, not as an absolute preventative guarantee.
What evidence cannot yet prove (limitations)
Important limitations in the evidence base:
- Lack of randomized trials testing Mirena specifically for primary prevention of endometrial cancer in average-risk women.
- Confounding in observational data (e.g., healthy-user effects, differences in health service contact, concurrent treatments) that may exaggerate or obscure true effects.
- Heterogeneity of populations and follow-up lengths in studies, which complicates pooled estimates of long-term protection.
- Safety trade-offs (see later), especially newer registry signals about breast cancer risk that need careful context.
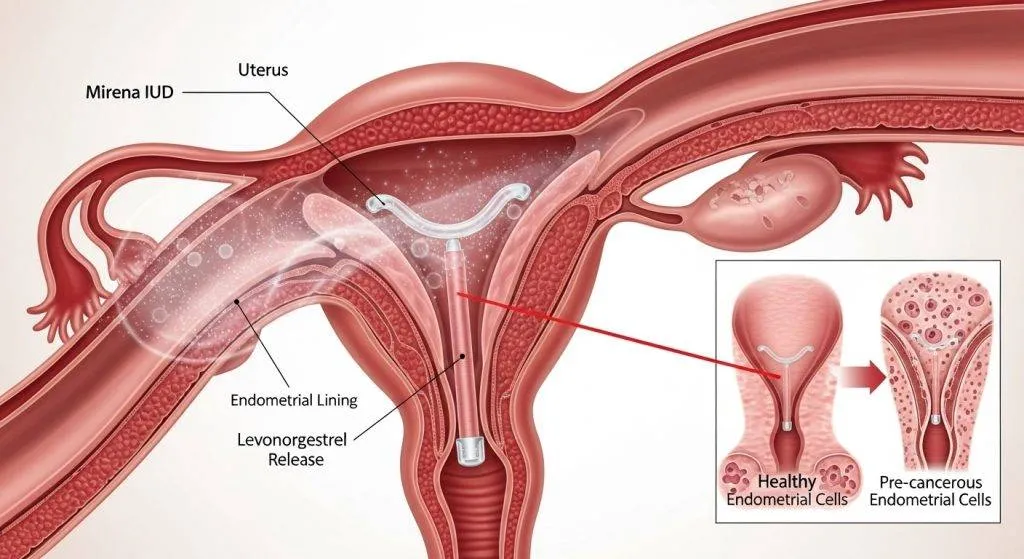
How Mirena affects the endometrium (mechanism of action)
Mirena’s protective association is rooted in its direct effects on the endometrial tissue. The biology explains why the device performs well for both bleeding control and reducing proliferative changes.
Endometrial thinning and atrophy: the biological effect
Levonorgestrel causes the glands and stroma of the endometrium to become inactive and thin. Cells that would otherwise proliferate under estrogen influence receive progestin signaling, which limits division and encourages a quiescent lining.
Counteracting estrogen’s effects: why balance matters
Endometrial cancer often arises when estrogen stimulation is unopposed by progesterone. Mirena supplies strong local progestin, restoring hormonal balance inside the uterus even when systemic factors (like obesity or anovulation) increase estrogen exposure.
Metaphor/analogy for patients
Think of the endometrium as a garden: estrogen is water and sunshine that make plants grow; Mirena acts like a gardener who trims and keeps growth in check so weeds (abnormal cells) don’t take over.
Mirena in therapeutic settings: hyperplasia & fertility-sparing care
Beyond prevention-framed benefits, Mirena is actively used to treat precancerous conditions and in carefully selected fertility-preserving strategies.
Use in endometrial hyperplasia (without atypia)
For simple or complex hyperplasia without atypia, LNG-IUS often reverses abnormal changes and controls bleeding. Many gynecologists prefer it over systemic progestins because of local potency and fewer systemic effects.
Hyperplasia with atypia and early cancer — cautious, specialist approach
When atypia is present or in early low-grade carcinomas, Mirena may be offered to women who wish to preserve fertility — but only after careful counseling, rigorous biopsy surveillance, and collaboration between gynecologic oncologists and reproductive specialists.
Fertility-sparing protocols in Spain: what specialists (doctors) do
In Spanish centres, specialists combine endometrial sampling, Mirena insertion, and close follow-up (regular imaging and biopsies). Decisions hinge on lesion grade, patient age, fertility desires, and adherence to follow-up plans.
Safety considerations & recent debates (what to discuss with your doctor)
Mirena’s uterine benefits come with a need to weigh systemic signals and side effects. A balanced discussion with your doctor should include both uterine advantages and potential broader risks.
Breast-cancer signals from registries: context and absolute risk
Some registry analyses reported a slight increase in breast cancer diagnoses among LNG-IUS users compared with non-users. Experts caution that absolute increases are small and influenced by age and prior hormone exposures. For women with a strong family history, this finding should inform, not dictate, decisions.
Balancing uterine benefits vs. systemic risks
Clinicians balance Mirena’s local endometrial protection and bleeding benefits against any marginal systemic signals. For many patients, uterine benefits and contraceptive convenience outweigh theoretical systemic concerns — but this is a personalized judgment.
Common side effects and rare complications (practical note)
Common early side effects include irregular bleeding and cramping; long-term effects often include lighter or absent periods. Rare complications include insertion-related infection or device expulsion. Contraindications (e.g., known uterine cancer, unexplained bleeding) mean a clinician must evaluate before insertion.
Who might benefit most from Mirena’s protective effect?
Mirena is particularly useful when local progestin action addresses the main risk drivers for a patient’s endometrium.
High-risk profiles: obesity, PCOS, long unopposed estrogen
Women with obesity, PCOS (Polycystic Ovary Syndrome), chronic anovulation, or prolonged estrogen exposure face higher endometrial cancer risk; Mirena can reduce endometrial proliferation for many in these groups.
Perimenopausal women and those on estrogen therapy
Perimenopausal women and those using systemic estrogen (e.g., for menopausal symptoms) may use Mirena to provide local progestin protection when indicated.
Who should NOT use Mirena (key contraindications)
Mirena is contraindicated with known or suspected uterine/cervical cancer, acute pelvic infection, or unexplained genital bleeding until diagnostic evaluation is complete.
Practical patient questions answered
Patients want clear, actionable answers to common concerns about Mirena’s protective role.
Two lines preview: Mirena lowers risk but doesn’t eliminate it; follow-up and symptom vigilance remain essential.
Does Mirena completely prevent endometrial cancer?
No — Mirena reduces risk for many, but it is not an absolute guarantee against cancer.
How long does protection last while the device is in place?
The endometrial effect persists while the device remains active (typical labeled duration varies by product, usually several years). Continued protection depends on continued presence and clinical circumstances.
Can Mirena replace surveillance or surgery?
Not for confirmed high-grade cancer; Mirena may be part of conservative management in selected cases but never replaces indicated surgical treatment for established high-risk disease.
What follow-up should I expect after insertion?
Expect an initial check, instructions on what symptoms to report, and routine gynecologic follow-up; for hyperplasia or fertility-sparing care, regular biopsies and imaging may be scheduled.
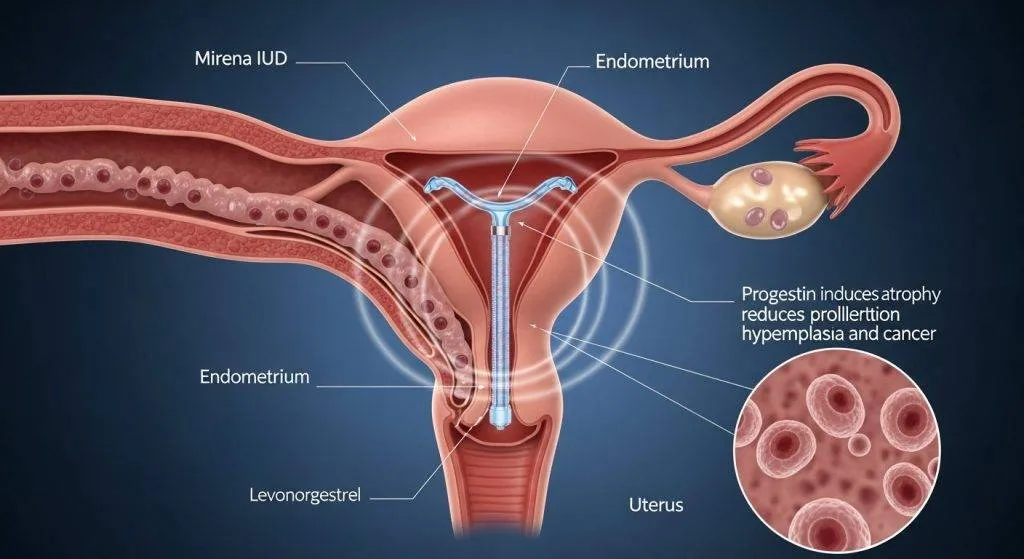
How doctors like Dr. Lucas in Spain approach Mirena and endometrial risk
Spanish clinicians contextualize Mirena within national guidance and local patient needs, emphasizing shared decision-making.
Regulatory and prescribing notes (AEMPS/CIMA)
Clinicians consult the AEMPS/CIMA product information for approved indications and safety details when prescribing Mirena in Spain.
Typical consultation flow: assessment, ultrasound, biopsy if needed
A pragmatic consultation often looks like this:
- Clinical history: bleeding pattern, risk factors (BMI, PCOS, family history), contraception needs.
- Pelvic ultrasound to assess endometrial thickness and uterine anatomy.
- If abnormal bleeding or thickened lining: office endometrial sampling (pipelle) or hysteroscopy + directed biopsy before placing Mirena.
- Shared decision on LNG-IUS vs alternatives, documentation of consent, and a follow-up plan (especially when used for hyperplasia or fertility-sparing care).
That stepwise approach is how Spanish gynecologists combine safety, diagnosis and treatment.
Mirena vs other hormonal options (brief comparison)
Mirena offers targeted local action; other options provide systemic protection but with different profiles.
Mirena vs oral progestins
Oral progestins work systemically and can be effective for hyperplasia, but they often cause systemic side effects and require strict adherence.
Mirena vs combined oral contraceptives
Combined pills lower endometrial cancer risk too, but Mirena excels in local, long-acting, low-systemic exposure and is preferred when endometrial atrophy is desired.
Why local hormone delivery matters
Local delivery means a potent uterine effect with fewer systemic hormonal symptoms — a key advantage for many patients.
FAQs
Does Mirena stop endometrial cancer completely?
No. It lowers risk for many patients but does not guarantee absolute prevention.
Can Mirena treat precancerous endometrial changes?
Yes — it is often effective for hyperplasia without atypia and may be part of conservative care for some select atypical cases under specialist supervision.
Is Mirena safe if I have a family history of breast cancer?
This requires individualized discussion. Recent data suggest a small breast-cancer signal in some registries; talk with your doctor about family history and alternatives.
How often should I be followed after Mirena insertion?
Routine checks occur after insertion and annually or as recommended; if Mirena is used for hyperplasia or fertility preservation, follow-up is more frequent and includes biopsies.
What should I do if I have abnormal bleeding with Mirena?
Contact your gynecologist. Unexplained bleeding needs assessment, which may include ultrasound and endometrial sampling to rule out pathology.
Conclusion
Mirena is a valuable tool: it reliably thins the endometrium and is associated with reduced endometrial cancer risk in many studies. However, it should be framed as risk reduction rather than absolute prevention. Discuss your individual risks and goals with a trusted gynecologist — clinicians like Dr. Lucas in Spain can help weigh benefits, alternatives, and monitoring plans.